Ribostamycin sulfate
Ribostamycin Sulfate
CAS: 53797-35-6
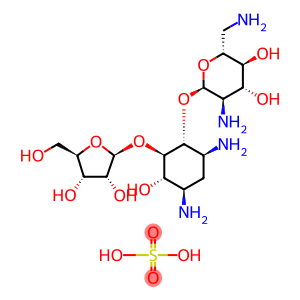
Molecular Formula: C17H22N4O10S
Ribostamycin sulfate - Names and Identifiers
| Name | Ribostamycin Sulfate |
| Synonyms | Ribomycine Ribostamin landamycine Landamycine Ribostamycin s Riboflavine sulfate Ribostamycin Sulfate Ribostamycin sulfate Rribostamycin sulfate Ribostamycin sulphate Ribose neoMycin sulfate SF 733 antibioic sulfate DTDP-GLUCOSE, DISODIUM SALT o-2,6-diamino-2,6-dideoxy-alpha-d-glucopyranosyl-(1-4)-o-(beta-d-streptamin Streptamine, O-2,6-diamino-2,6-dideoxy-alpha-D-glucopyranosyl-(1-4)-O-(beta-D-ribofuranosyl-(1-5))-2-deoxy-, sulfate |
| CAS | 53797-35-6 |
| EINECS | 258-783-1 |
| InChI | InChI=1/C17H34N4O10.H2O4S/c18-2-6-10(24)12(26)8(21)16(28-6)30-14-5(20)1-4(19)9(23)15(14)31-17-13(27)11(25)7(3-22)29-17;1-5(2,3)4/h4-17,22-27H,1-3,18-21H2;(H2,1,2,3,4)/t4-,5+,6-,7-,8-,9?,10-,11-,12-,13-,14-,15-,16-,17+;/m1./s1 |
Ribostamycin sulfate - Physico-chemical Properties
| Molecular Formula | C17H22N4O10S |
| Molar Mass | 474.44238 |
| Melting Point | 175-180 C |
| Specific Rotation(α) | D20 +39° (c = 1) |
| Flash Point | >110°(230°F) |
| Water Solubility | Soluble in water |
| Solubility | H2O: soluble50mg/mL |
| Appearance | powder |
| Color | White to Off-White |
| Merck | 14,8206 |
| Storage Condition | Inert atmosphere,2-8°C |
| Stability | Hygroscopic |
| Use | For the treatment of bronchitis caused by sensitive bacteria, Pneumonia, pleurisy, lymphadenitis, cholecystitis, furunculosis, osteomyelitis, urinary tract infection |
Ribostamycin sulfate - Risk and Safety
| Risk Codes | R61 - May cause harm to the unborn child R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/38 - Irritating to eyes and skin. |
| Safety Description | S53 - Avoid exposure - obtain special instructions before use. S22 - Do not breathe dust. S36/37/39 - Wear suitable protective clothing, gloves and eye/face protection. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) S37/39 - Wear suitable gloves and eye/face protection S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. |
| WGK Germany | 3 |
| RTECS | WK2300000 |
| Toxicity | LD50 i.v. in mice: 225 mg/kg (Shomura) |
Ribostamycin sulfate - Reference
| Reference Show more | 1. Wei Qinglan, Zhang Jipei, Ren Tao, et al. Drug Sensitivity Analysis of Escherichia coli from waterfowl in Guangdong province [J]. Heilongjiang Journal of Animal Husbandry and Veterinary Medicine, 2015, 000(012):180-181. 2. Liu Minfang, Cao mengrui, Li Rongxu, etc. Isolation, identification and drug sensitivity analysis of salmonella from waterfowl in some areas of Guangdong province [J]. Heilongjiang Journal of Animal Science and Veterinary Medicine, 2018(10). |
Ribostamycin sulfate - Reference Information
| overview | aminoglycoside antibiotics are another class of widely used antibiotics besides β-lactam antibiotics (I. e. penicillins and cephalosins). In 1944, streptomycin was discovered from the actinomycete culture solution, which was the first generation of aminoglycoside antibiotics. Later, neomycin, gentamicin, paromomycin, kanamycin, and dideoxykanamycin and amikacin were discovered. The above drugs are relatively toxic to the auditory nerve and kidney. Bacteria with R factor can passivate these antibiotics. In order to solve the above problems, pharmaceutical workers are committed to the research of a new generation of aminoglycoside antibiotics with high efficiency and low toxicity. Promising antibiotics are salamicin, fatimycin, cisosamycin, ethylcisosamycin and ribosomycin, of which ribosamycin sulfate is a kind of clinical application with good curative effect and low toxicity. Ribomycin sulfate is an aminoglycoside antibiotic. The antibacterial spectrum is similar to kanamycin, and its toxicity is lower than that of kanamycin. It has good effects on Escherichia coli, Klebsiella, Proteus, Staphylococcus, Neisseria gonorrhoeae, and Neisseria meningitidis. The second part of the 2005 edition of the Pharmacopoeia of the People's Republic of China contains ribosomycin sulfate and ribosomycin sulfate for injection, and the "National Drug Standards" (Volume 2 of the National Standard for the Rise of Local Standards for Chemical Drugs) contains ribosomycin sulfate injection. The Japanese Pharmacy also accepted this variety. After entering the human body, this product can bind to the 30S subunit of the bacterial ribosome and inhibit the synthesis of protein by bacteria. It is clinically used for respiratory tract, abdominal cavity, thoracic cavity, urinary tract, skin and soft tissue, bone tissue, eye, ear and nose infections caused by sensitive gram-negative bacilli. This product has good antibacterial activity against Escherichia coli, Klebsiella, Proteus and other Enterobacteriaceae bacteria, has weak effect on Mycobacterium tuberculosis and Streptococcus, and has no effect on Pseudomonas aeruginosa and anaerobic bacteria. The bacteria are cross-resistant to the product and kanamycin. In clinical practice, the antibacterial spectrum of this product is narrow when administered alone, and bacterial resistance is prone to occur. The combined administration of ribosomycin sulfate and low molecular dextran can aggravate the damage to the kidneys. |
| action | ribomycin sulfate is an aminoglycoside antibiotic obtained by extracting the fermentation broth of the strain obtained by mutagenesis screening of domestic streptomycin 3719. Ribomycin sulfate is an aminoglycoside antibiotic with a wide antibacterial spectrum. It has strong antibacterial activity against various Gram-positive and Gram-negative bacteria. It is resistant to Staphylococcus aureus, Streptococcus, Pneumococcus, Escherichia coli, Shigella and other sensitive bacterial infections, such as sepsis, bronchitis, pulmonary purulent, empyema, biliary tract infection, peritonitis and urinary system infection, etc, there is no cross-resistance with penicillin G, tetracycline, ampicillin or o-clopicillin, and cross-resistance with carramycin, good absorption, high blood concentration, and lower toxicity than other aminoglycoside antibiotics. |
| preparation | a preparation method of high-purity ribonomycin sulfate, comprising the following steps: step 1: adjust the ribonomycin sulfate resolution solution to be acidic and dilute it; Step 2: analyze the ribonomycin sulfate hydraulically into an off-cross column filled with resin, then wash with deionized water, adsorb, and sample for optical rotation; step 3: Continue washing with ammonia water, take samples to measure the rotation until the rotation is "0", and stop washing; Step 4: Concentrate the ribosomycin sulfate parsing solution; Step 5: Take the ribosomycin sulfate concentrated solution into salt and remove it to obtain ribosomycin sulfate decolorizing solution, and finally spray drying to obtain the finished product. Among them, in step one, sulfuric acid is used to control the pH value of the ribosomycin sulfate resolution solution at 5.0, and purified water is used to dilute the ribosomycin sulfate resolution solution to 18 μ/ml. The concentration of sulfuric acid is 10%, and the pH value of ribomycin sulfate resolution solution is adjusted at 15 ℃. In step 2, the ribosomycin sulfate analytical solution is pressed into the resin-filled separation column at a flow rate of 180L/h, and then washed with deionized water at a flow rate of 180L/h for 7 hours, adsorbed for 3 hours, and sampled every 1 hour to measure the rotation. In step 3, wash with 0.1mol/L ammonia water at a flow rate of 190L/h for 70h, then adjust the flow rate of ammonia water to 90L/h for washing, sample and measure optical rotation. when optical rotation is less than 0.4, add 1.5mol/L ammonia water to continue washing and control the flow rate to 280L/h until optical rotation is "0". In step four, the ribosomycin sulfate resolution liquid is concentrated through the nanofiltration membrane, and the temperature is controlled below 40 degrees C. In step 4, secondary concentration is carried out through a steam membrane device, and the vacuum degree ≤-0.08Mpa, the temperature ≤ 65 ℃, and the flow rate 400L/h are controlled. |
| preparation method | a lyophilized powder injection of ribosomycin sulfate for injection, comprising: ribosomycin sulfate 1000g; Dissolve ribosomycin sulfate to 500g/L with 2000mL of water for injection; The preparation method includes the following steps:(1) preparing a semi-finished product of ribosomycin sulfate: accurately weigh the prescription amount of ribosomycin sulfate, add it to the prescription amount of water for injection, stir and dissolve until the solution is clarified; The activated carbon is added for stirring and adsorption for 30 minutes, and the dosage of activated carbon is 0.1% g/mL; The activated carbon is removed and filtered by 0.45 μm and 0.22 μm microporous filter membrane, and the obtained filtrate is ribocamycin sulfate semi-finished product. (2) Freeze-drying the ribomycin sulfate semi-finished product, the freeze-drying includes the following three stages: pre-freezing stage: first reduce the temperature of the freeze dryer layer to 0°C, and put the ribomycin sulfate semi-finished product into the freeze dryer, Keep for 30 minutes, then drop to -40°C for 1 hour, keep for 2 hours; sublimation drying stage: the temperature of the plate layer is raised at a rate of 4°C/hour to -5°C ~ 0°C, and keep it for 3 hours; analytical drying stage: the temperature of the plate layer is raised at a rate of 8 ℃/hour to 35 ℃, and kept for 3 hours to obtain ribomycin sulfate freeze-dried powder injection. |
| traits | carramycin sulfate is white or white-like powder. |
| use | carramycin sulfate is a broad-spectrum antibiotic, which has antibacterial effect on gram-positive and negative bacteria. it is used for infections caused by Escherichia coli, aerogenes, enteritidis, dysentery bacillus, enterococcus, drug-resistant staphylococcus, etc. for the treatment of bronchitis, pneumonia, pleurisy, lymphadenitis, cholecystitis, boils, osteomyelitis, urinary tract infection, etc. caused by sensitive bacteria |
Last Update:2024-04-09 20:52:54
Supplier List
Spot supply
Product Name: Ribostamycin sulfate Visit Supplier Webpage Request for quotationCAS: 53797-35-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Framycetin Sulfate Impurity 11 Sulfate Visit Supplier Webpage Request for quotationCAS: 53797-35-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Ribostamycin Sulfate Request for quotation
CAS: 53797-35-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 53797-35-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Ribostamycin sulfate Request for quotation
CAS: 53797-35-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 53797-35-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Ribostamycin Sulfate salt Visit Supplier Webpage Request for quotationCAS: 53797-35-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Ribostamycin sulfate Visit Supplier Webpage Request for quotationCAS: 53797-35-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Framycetin Sulfate Impurity 11 Sulfate Visit Supplier Webpage Request for quotationCAS: 53797-35-6
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: Ribostamycin Sulfate Request for quotation
CAS: 53797-35-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 53797-35-6
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Ribostamycin sulfate Request for quotation
CAS: 53797-35-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 53797-35-6
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Multiple Specifications
Product Name: Ribostamycin Sulfate salt Visit Supplier Webpage Request for quotationCAS: 53797-35-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




